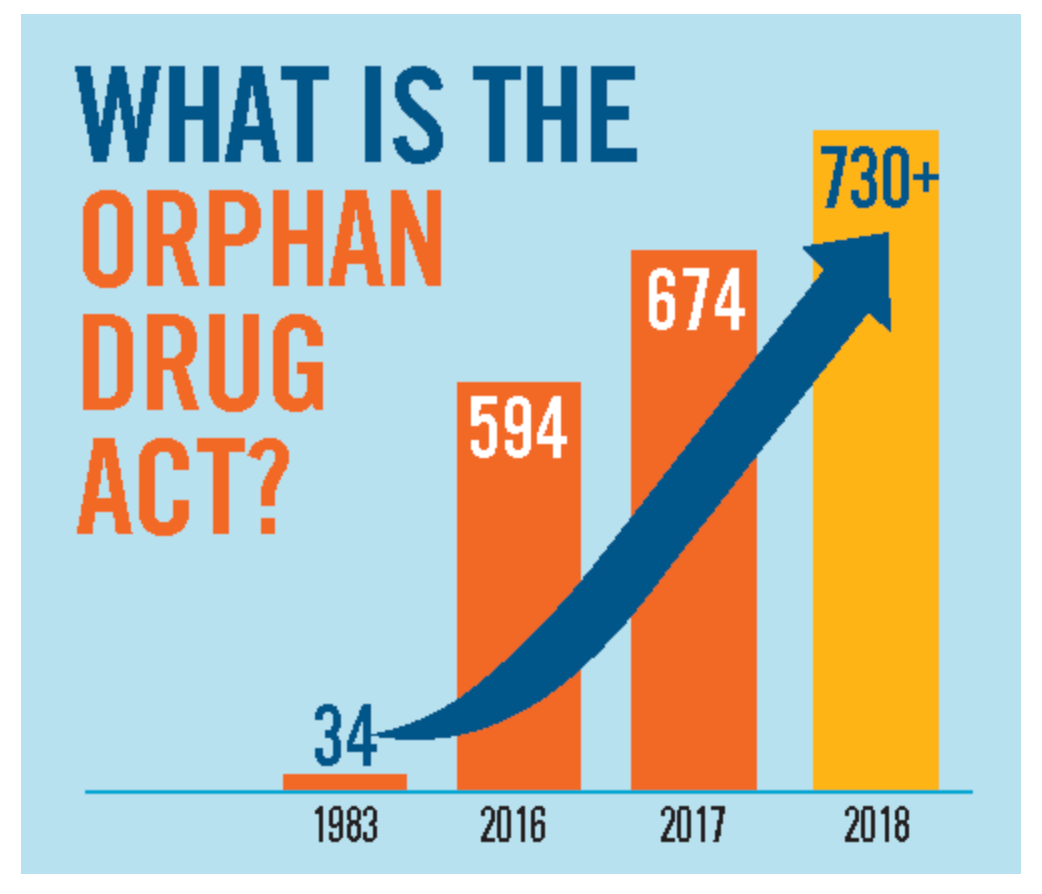
FDA Proposes New Approval Pathway for Ultra-Rare Disease Therapies
Feb 26, 2026
The MAHA Report
We are dedicated to providing accurate information about the MAHA movement including articles on the key issues such as public health, exercise, censorship, media bias, homelessness, drug addiction, and the environment
The MAHA report is an affiliate of MAHA Action Inc.
Learn more at: https://www.mahaaction.com/
We are dedicated to providing accurate information about the MAHA movement including articles on the key issues such as public health, exercise, censorship, media bias, homelessness, drug addiction, and the environment
The MAHA report is an affiliate of MAHA Action Inc.
Learn more at: https://www.mahaaction.com/Listen on
Substack App
Apple Podcasts
Spotify
RSS Feed
Recent Episodes