Weekly Wins: HHS Hosts Historic Women’s Health Conference, the MAHA Institute Holds Vaccine Injuries Round Table, a New System to Track Adverse Health Events, and More
In another landmark week for the MAHA movement, the FDA unveiled a centralized dashboard for adverse health events, the MAHA Institute hosted a stellar summit on vaccine safety, Kansas advanced key legislation for student wellness, and Tennessee ditched dyes.
But first….we invite you to watch Caitlin Sinclair’s light and lively MAHA Minute, available every Friday across MAHA Action’s social media channels.
HHS Hosts Triumphant Women’s Health Conference
From March 11 to 13, HHS hosted the National Conference on Women’s Health in Washington, DC. This historic event convened experts, clinicians, policymakers, and advocates to discuss breakthroughs in areas such as hormonal health, menopause, chronic disease prevention, the female reproductive cycle, and innovative approaches to disease prevention and treatment across women’s lifespans.
[Look for more coverage of this event in The MAHA Report in the coming days.]
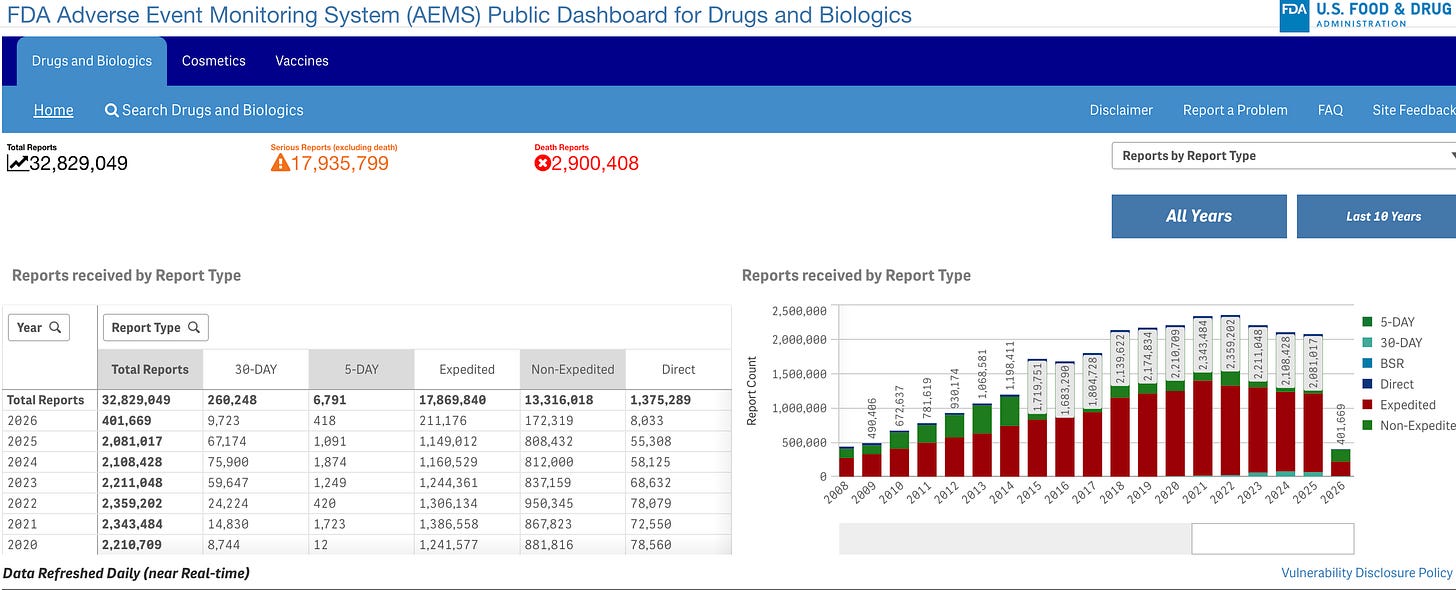
FDA Creates New Adverse Event Monitoring System
The FDA launched the Adverse Event Monitoring System (AEMS), a centralized dashboard designed to provide “radical transparency” by consolidating safety reports for drugs, vaccines, cosmetics, and more. By May 2026, the agency plans to migrate all historical data and integrate real-time reporting for all FDA-regulated products into this single interface.
Beyond improving data accessibility, the transition is expected to modernize the agency’s infrastructure and reduce operational costs by $120 million over the next five years.
The AEMS platform will consolidate several legacy databases into a single interface, initially replacing FAERS (drugs and cosmetics), VAERS (vaccines), and AERS (animal products). By May 2026, the transition will include MAUDE for medical devices, HFCS for human foods and dietary supplements, and CTPAE for tobacco products, effectively unifying the FDA’s safety monitoring for all regulated goods.
MAHA Institute Round Table Gives Vaccine Injuries Long-Overdue Close-Up
On March 9, the MAHA Institute hosted the MEVI Round Table in Washington, D.C., focusing on the origins of and solutions to what the Institute calls ‘the Massive Epidemic of Vaccine Injury.’
MAHA Institute President, Mark Gorton, proposed adopting the acronym MEVI into the public lexicon to explicitly link the rise in chronic illnesses—including allergies, autoimmune diseases, and neurodevelopmental conditions like autism—to unsafe vaccination practices. The event combined scientific analysis with personal testimonials advocating for policy changes aimed at addressing these health trends.
The round table also emphasized the growing legislative and social movement toward medical freedom, as covered by The MAHA Report, here.
Mary Holland, President of Children’s Health Defense, cited polling data suggesting that a supermajority of Americans support the right to make their own medical decisions, while advocate Del Bigtree argued that true national freedom is incompatible with bureaucratic interference in personal health choices. Together, the speakers framed the issue as both a public health crisis and a fundamental struggle for individual bodily autonomy.
New Law Helps Make Kansas Healthy Again
The Kansas Senate passed SB 339, a bill requiring public school districts to provide a minimum of 30 minutes of daily, mostly unstructured recess for students in kindergarten through eighth grade.
Tennessee Ditches Dyes
In Tennessee, HB1853 passed in the House. Last month, SB 2423, the Senate version, also passed. The bill prohibits schools from serving students foods containing artificial food dyes in their nutrition programs.
Dignity for Organ Donors
CMS issued new guidance aimed at restoring public trust and ensuring that organ donation and procurement processes are handled with dignity and transparency.
These safeguards reinforce a requirement that hospitals provide full medical care to all patients regardless of their potential donor status and prohibit Organ Procurement Organizations (OPOs) from pressuring grieving families or influencing the timing of life support withdrawal. By requiring surveyors to cite noncompliance even after it has been corrected, CMS is heightening accountability to protect patients and families from coercive practices.
“Americans’ trust in physicians and hospitals decreased more than 40% between 2020 and 2024,” noted CMS Administrator Dr. Mehmet Oz. “In this administration, we’re committed to earning it back. We’re restoring trust by promoting transparency, strengthening accountability, and respecting the dignity of patients and their families.”







From start to finish on Monday, the MEVI summit meeting was the most powerful and impactful day I've ever been apart of! It was exactly what an entire generation of parents and children have been waiting for - the truth!
Labels should be required on food that has been sprayed with glyphosate. Citric acid (mold) should be eliminated. “Natural flavors” exposed (MSG).
Also scented products are toxic. They are poisoning us. They’re putting these toxic fragrances in everything including laundry products, air fresheners, trash bags, etc. Air “fresheners” are in hospitals, medical office, grocery stores, GYM’s,. They should be labeled air poisoners.