Why FDA and HHS Are Rethinking Food Safety
For 30 years, the federal government allowed the food industry to determine which ingredients are safe. That’s about to change.
By Amy Sapola, Pharm.D., Contributor, The MAHA Report
Turn over almost any package of ultra-processed food, and you will find a long list of ingredients that are difficult to pronounce and definitely not something our great-grandparents would recognize. Yet the assumption remains: if it is in the food supply, it must have been reviewed by the U.S. Food and Drug Administration (FDA) for safety…right?
In many cases, this is simply not true.
For decades, thousands of chemical substances have entered the American food supply through a regulatory loophole known as “generally recognized as safe,” or GRAS. They entered without formal review or mandatory disclosure to the FDA – and, in some cases, without any publicly available safety data.
Even more striking is what is unknown. Because companies are not required to notify the government of self-affirmed GRAS determinations, there is no complete accounting of how many such substances are currently in the U.S. food supply.
“The GRAS loophole has allowed food companies to decide for themselves whether a chemical is safe, without notifying the FDA,” said HHS Secretary Robert F. Kennedy Jr. “That is a clear conflict of interest, and it undermines public trust.”
Kennedy added, “We are going to fix the GRAS system so that chemicals cannot be added to food without oversight. The American people deserve to know what is in their food and that it has been independently evaluated for safety.”
Watching GRAS Grow
The modern GRAS framework began with the Food Additives Amendment of 1958, when Congress created a premarket approval system for new food additives while deliberately exempting substances already widely recognized as safe. At the time, this category was relatively small and logical, covering ingredients like salt, vinegar, spices, and oils with long histories of human use in cooking.
Early efforts by the FDA in the 1960s and 1970s sought to formalize GRAS ingredients by publishing lists and a comprehensive review program that evaluated hundreds of substances already in the food supply. However, regulators acknowledged a fundamental limitation: it was impracticable to identify and evaluate every substance and every use as the food system rapidly industrialized.
By the 1970s and 1980s, the scale of the problem had grown. The FDA began issuing formal “GRAS affirmation” regulations, but the process was slow, resource-intensive, and unable to keep pace with the expanding number of ingredients and novel uses. During this time, roughly 700 to 1,000 substances had been formally recognized or reviewed as GRAS, but this represented only a portion of those actually in use. In practice, companies increasingly relied on independent scientific experts to make safety determinations, as FDA oversight struggled to keep pace.
The most consequential shift came in 1997, when the FDA proposed replacing the GRAS process with a voluntary notification system. This change did not come from a single political directive but from a convergence of regulatory pressure, industry influence, and administrative reality. The FDA faced significant backlog and resource constraints, while the food and ingredient industries were pushing for a more efficient pathway to bring products to market. Trade associations representing food manufacturers, chemical suppliers, and ingredient producers had long advocated for reducing their burden of bringing new products to market, arguing that existing scientific expertise within industry could support safety determinations more efficiently than waiting for the FDA.
.Over time, this contributed to a model in which safety assessments became increasingly privatized, with companies convening their own “expert” panels and generating their own data that was not required to be shared with the FDA.
Under the 1997 proposal, companies could self-determine that a substance was GRAS and were encouraged, but not required, to notify the FDA. This shift was framed as a modernization effort, but it fundamentally altered the structure of oversight. From that point forward, GRAS became a system in which new substances could enter the food supply without mandatory FDA review or ANY disclosure of safety data.
Since the late 1990s, this decentralized model has defined the modern GRAS landscape. The FDA has received and reviewed over 1,000 voluntary GRAS notices, creating a partial public record of substances evaluated through the notification pathway. However, independent analyses and policy reviews suggest that thousands more self-affirmed GRAS determinations exist outside of FDA oversight, meaning the total number of substances in use is not fully known.
What began in 1958 as a narrow, common-sense exemption for household ingredients used in cooking has grown into a way for industry to skirt the FDA and introduce thousands of new chemicals into the food supply for use in ultra-processed foods.
The Dose Makes the Poison — or Does It?
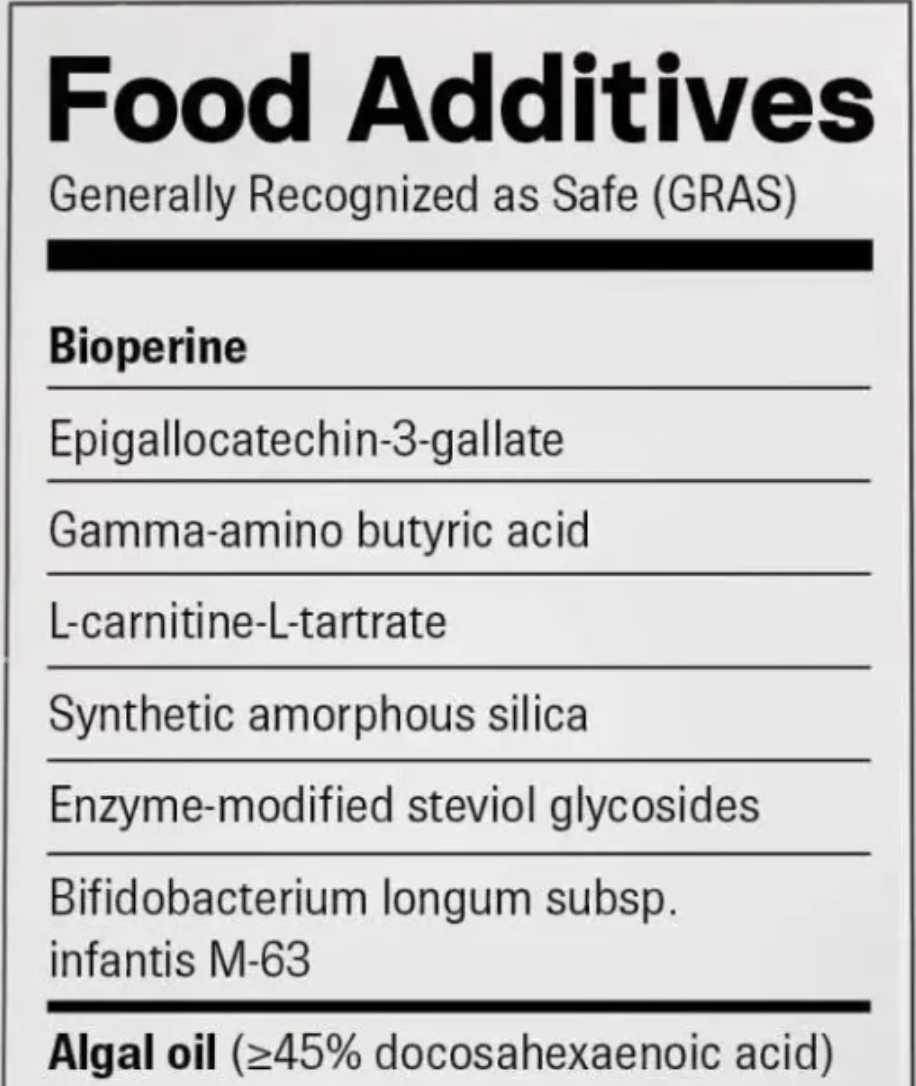
One of the clearest ways to understand the limitations of the GRAS framework is to look at how these substances actually appear in our food supply. Some of the least transparent categories include flavoring agents, such as natural and artificial flavors, as well as processing aids used during manufacturing, such as hexane, ammonium hydroxide, and dimethylpolysiloxane, which often do not even appear on labels.
While terms such as “natural flavor” may sound familiar and safe, they often refer to complex mixtures of dozens or even hundreds of chemical compounds. Much of the underlying safety data is proprietary and not publicly available, yet these substances are allowed in the food supply under GRAS.
A second layer of complexity lies in how GRAS is applied.
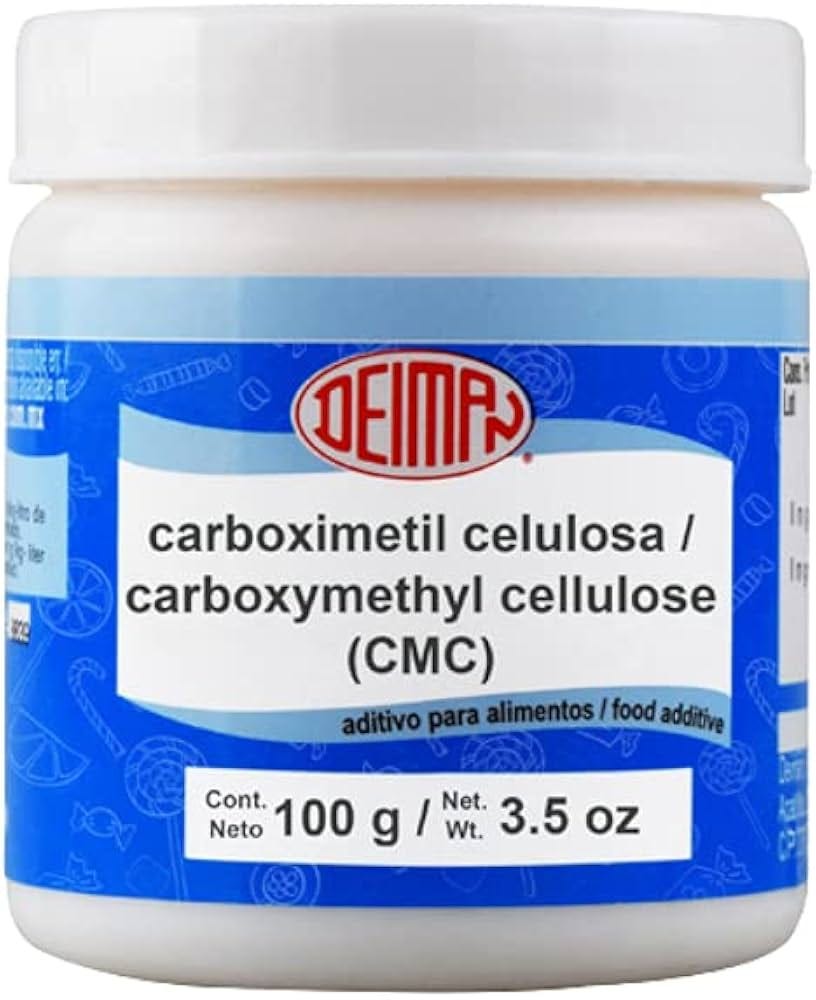
Carboxymethylcellulose (CMC), a synthetic thickener derived from plant fiber, may be considered safe when used in small amounts in a single product. But in reality, it is consumed across many ultra-processed foods, often multiple times per day, and in combination with other emulsifiers such as lecithin, polysorbate 60, and polysorbate 80. It is important to note that GRAS applies to a specific use in a specific food, but not to chronic and potentially cumulative exposure across a person’s life.
Emerging research suggests that some emulsifiers, including CMC, may alter the gut microbiome and intestinal lining with chronic exposure, effects not captured in traditional GRAS evaluations focused on acute toxicity. The result is a system that evaluates ingredients in isolation, whereas in reality, Americans who consume ultra-processed foods, now accounting for 60%-70% of calories in the “Standard American Diet,” are exposed to complex mixtures daily.
Why Reform Is Now on the Table
Secretary Kennedy has made reforming the GRAS loophole a priority, with recent statements and policy signals from the HHS and FDA indicating that efforts are underway to reevaluate and strengthen oversight of the GRAS pathway.
While formal rule changes have not yet been implemented, the agencies have outlined plans to increase transparency, modernize safety standards, and consider reforms to the current self-affirmation process that underpins the GRAS loophole.
At the same time, industry pushback has been significant. Trade groups argue that mandatory notification could exceed FDA authority, create regulatory bottlenecks, and disrupt supply chains. Others raise concerns about whether reforms will require retroactive review of ingredients used for decades.
These tensions highlight what is at stake. Not just regulatory efficiency, but the fundamental question of who determines what is safe.
A Defining MAHA Priority
Closing the GRAS loophole is about restoring a fundamental standard: that safety determinations in the American food supply are transparent, independently evaluated, and grounded in modern, gold-standard science.
For clinicians and government leaders alike, this represents a rare opportunity to move upstream and acknowledge that the ingredients in our food supply are foundational drivers of health across a lifespan. It is an opportunity to realign the system itself with that reality, prioritizing long-term health over short-term convenience.
For families, this moment invites a more critical question: what does “safe” truly mean? And until that standard is strengthened, it reinforces the importance of choosing whole, minimally processed foods whenever possible.
The HHS and the FDA are now beginning the important work of restoring accountability in the American food system and rebuilding trust in the institutions responsible for protecting public health. This moment demands more than incremental change. It requires a clear commitment to closing long-standing loopholes and ensuring that industry interests do not stall reform or rely on anything other than transparent, gold-standard science.
Secretary Kennedy and team are seizing the opportunity to finally close the gap between what is assumed to be safe and what is proven to be safe, and to put public health where it belongs: first.








It would take research to finally prove it however I believe it would reveal the massive number of chemicals in our food would show it hinders a humans ability to think critically.
This alone is reason to ban most if not all chemicals/substances not found in whole foods.
Glad to see this happening. Perhaps having an accredited 3rd party review GRAS materials would relieve the burden on FDA to get the job done. (Somewhat like the Organic Materials Review Institute, OMRI, as the gold standard for materials used in USDA certified-organic agriculture).
Moreover, as a veterinarian, I hope that pets and livestock are also addressed by MAHA since our lives are so interwoven with our pets and livestock.
By the way, the FDA Center for Veterinary Medicine (FDA CVM) keeps its own GRAS listing, which began around 2012 and reflects many substances which are definitely not "traditional" food/feed ingredients.. You can see the entire list here with hyperlinks to FDA respond see to each ingredient:
https://www.fda.gov/animal-veterinary/generally-recognized-safe-gras-notification-program/current-animal-food-gras-notices-inventory
Unfortunately, innocent entries like traditional berberine (derived from the plant Berberis vulgaris or Hydrastis canadensis) was raked over the coals by FDA CVM and withdrawn, whereas other novel foods/feeds that are clearly shown to be from genetically engineered materials got a "no objection" letter from FDA CVM. What???
The original 1958 list that showed plants and plant-derivatives (essential oils, spices, seasonings) ought to be incorporated into the FDA CVM GRAS list for animals. Doing so would also help natural substances be used on livestock without penalty by FDA inspectors on organic farms. Unfortunately many natural substances found and used on USDA certified-organic Grade A dairy farms are penalized on standard FDA inspection. This needs to change for MAHA friendly agriculture to flourish.