Why Are American Fertility Rates Declining – and What Can Be Done About It?
New platforms like Oova and Mira show metabolic dysfunction is a piece of the puzzle
“When my uncle was president, the fertility rate in this country was 3.5 percent. Today, it is 1.6 percent. The replacement rate, in other words, the amount of fertility that you need in order to keep your population even, is 2.1 percent. We are below replacement right now. That is a national security threat to our country . . . .”
– Robert F. Kennedy Jr.
The latest data from the Centers for Disease Control and Prevention (CDC) show that both America’s birth and fertility rates remain near historic lows. In 2024, the U.S. recorded roughly 700,000 fewer births than in 2007, the recent high point for total annual births.
But those numbers tell only part of the story. While 2007 marked the modern peak in the total number of babies born, America’s true fertility peak, measured by the average number of children women had, occurred during the 1950’s baby boom era, when the birth rate was 3.5 children per woman, compared with 2.1 in 2007.
Why is this happening?
Most headlines point to economics or delayed family planning. Both matter, but they mask a deeper, more urgent contributor, the menstrual cycle itself.
The Hidden Shift in Hormonal Patterns
For decades, the fertility conversation has largely focused on one variable: age. We all know American women are giving birth later, with today’s average nearly 30 years old, up from 1960, when it averaged close to 25. But a far bigger and more underreported story may be unfolding beneath the surface.
Later maternal age is only one piece of the puzzle. Metabolic dysfunction, chronic stress, poor sleep, and rising exposure to endocrine-disrupting chemicals like phthalates and bisphenols are now adding up in ways that may be quietly reshaping reproductive health. While we lack long-term hormone data needed to make accurate comparisons with previous generations, new cycle-tracking technologies are exposing patterns that much of medicine is currently overlooking.
Across full menstrual cycles, more women are showing irregular or incomplete ovulation, subtle but clinically important progesterone deficiencies, and surprising cycle-to-cycle hormonal instability even when standard bloodwork appears “normal.” The emerging story is not just about hormone levels being too high or too low. It may be about something even more consequential: growing disruption in the communication between the brain and the ovaries. And when that signaling becomes less stable, fertility itself becomes increasingly fragile.
Why Our Current Fertility Tools May Be Missing the Bigger Picture
In most medical settings, fertility is still often evaluated through a handful of one-time blood tests, typically checking estrogen early in the cycle and progesterone after suspected ovulation. These tests can offer helpful clues, but they are just snapshots: a single moment used to represent an entire month of hormonal activity.
That approach may miss far more than many women realize.
Hormones are not static. They rise, fall, and interact in carefully timed patterns throughout the month, and for women with irregular cycles, delayed ovulation, or subtle hormonal dysfunction, one “normal” lab result may not tell the full story. Increasingly, newer research suggests that fertility may be far more dynamic and far less predictable than conventional medical textbooks teach.
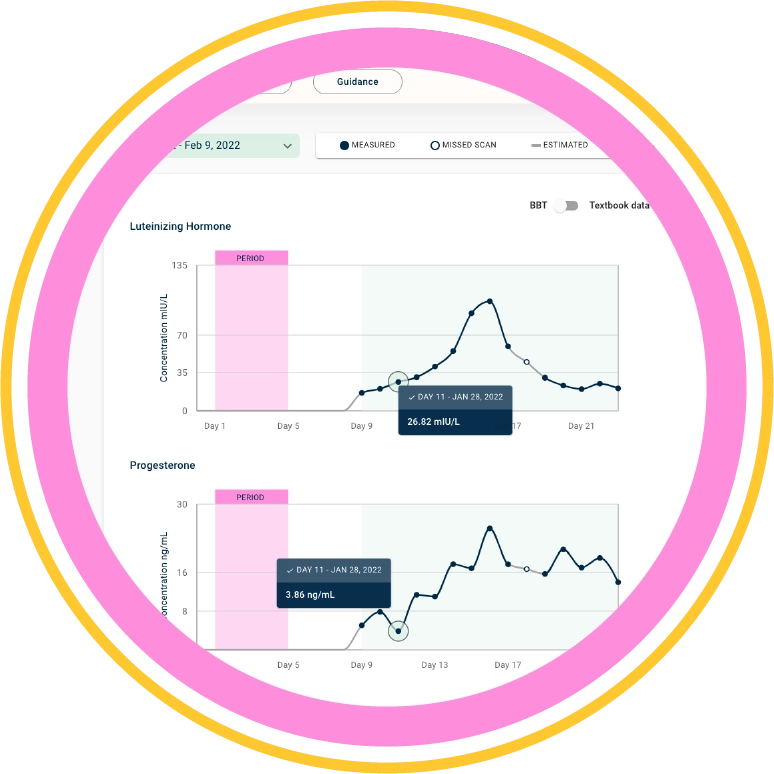
This is where newer at-home hormone monitoring tools like Oova and Mira are beginning to reshape the conversation. By using daily urine testing throughout the cycle, these platforms allow women to track changing hormone patterns over time rather than relying on a single blood draw. In simple terms, they may help answer a bigger question: not just whether hormones are present, but whether the entire system is functioning optimally.
Oova’s large-scale cycle research found that nearly one in five women experienced a luteinizing hormone (LH) surge, the hormonal signal that typically precedes ovulation, without confirmed ovulation. More than half of ovulatory cycles showed lower-than-expected progesterone during the luteal phase, a key hormone for implantation and early pregnancy support. Perhaps the most important finding was that hormone patterns varied significantly from cycle to cycle, challenging the familiar textbook image of the menstrual cycle as a smooth, predictable 28-day process.
Mira’s research reinforces a similar message. By tracking multiple hormones across the full cycle, including LH, estrogen, progesterone metabolites, and follicle-stimulating hormone (FSH), the data suggest that important disruptions in hormone timing, coordination, and rhythm may easily go undetected when fertility is reduced to isolated lab values alone.
The takeaway is both simple and profound: fertility may not be as straightforward as “you ovulated” or “your labs are normal.”
The menstrual cycle can be thought of as a dynamic vital sign, much like blood pressure or heart rate, because it often reflects what is happening in the body as a whole. Just as blood pressure can change in response to stress, diet, sleep, or illness, menstrual cycles can also shift in response to sleep quality, stress levels, nutrition, metabolic health, age, and environmental exposures. Some month-to-month variation is completely normal.
But when cycles become increasingly irregular, ovulation becomes inconsistent, or hormonal patterns change significantly, it could be an early signal that broader aspects of health, such as hormone balance, metabolic function, or overall physiologic resilience, deserve closer attention.
A System Under Strain
Emerging hormone research is beginning to confirm what MAHA has long warned: fertility cannot be separated from the broader metabolic and environmental conditions shaping modern health.
Reproductive health is deeply influenced by blood sugar regulation, insulin sensitivity, nutrition, sleep, chronic stress, and exposure to endocrine-disrupting chemicals.
That reality is especially relevant in a country where more than 40 percent of U.S. adults are living with obesity, according to the CDC. This is not simply about weight; it is about metabolic dysfunction, inflammation, and hormonal disruption that can directly affect ovulation, progesterone, and overall reproductive resilience. Research suggests women with obesity may face two to three times the risk of infertility related to ovulatory disorders.
The rise of GLP-1 medications such as semaglutide and tirzepatide is adding a new and rapidly evolving layer to the fertility conversation. Originally developed for diabetes and now widely used for weight loss, these drugs can improve insulin resistance, reduce inflammation, and, in some women, particularly those with obesity or PCOS, may help restore more regular ovulation by improving underlying metabolic health. At the same time, rapid weight loss, nutritional deficiencies, or unplanned conception during treatment may introduce significant risks if not carefully monitored, and long-term fertility-specific data are still limited.
GLP-1s may offer meaningful benefits for some individuals, particularly when metabolic dysfunction is playing a significant role in hormonal or reproductive challenges. But on a population level, fertility is unlikely to be restored by medication alone. The rapid rise of GLP-1s may instead highlight a deeper truth: when metabolic health improves, reproductive health often improves alongside it. The larger opportunity at the policy level is to address the root-cause systemic drivers affecting metabolic health, such as the quality of nutrition, food systems, and environmental exposures, which contribute to both chronic disease and fertility decline.
What Needs to Change
If there is a single takeaway from this story, it is this: women’s hormonal health is not static. It is rhythmic, adaptive, and influenced by the world women live in, from metabolic health and nutrition to sleep, stress, and environmental exposures.
The path forward is clear: Move beyond isolated lab snapshots and toward longitudinal hormone tracking that reflects the true complexity of female physiology. Expand fertility care beyond ovulation prediction alone to include metabolic resilience, blood sugar regulation, nutrient optimization, toxin reduction, and whole-person lifestyle medicine. Invest in research that finally studies women’s bodies the way they actually function, across full cycles, across decades, and across major hormonal transitions.
Most importantly, this moment offers an opportunity to shift from reactive fertility care to proactive reproductive health.
And while this story has focused primarily on women’s health, fertility is never solely a woman’s issue. Male fertility matters, and mounting research suggests that many of the same upstream forces, including metabolic dysfunction, ultra-processed diets, endocrine-disrupting chemicals, chronic stress, poor sleep, and environmental toxins, also contribute to declining sperm quality and lower testosterone levels in men. The health of future generations depends on both sides of the equation, and any serious fertility conversation must ultimately address reproductive resilience as a shared biological and societal priority.
For too long, women have been given fragmented answers inside fragmented systems. But if declining fertility is, in part, a reflection of broader upstream dysfunction, then improving fertility may also become one of the clearest measures of whether we are truly improving overall human health.
The fertility story does not have to end in decline. This moment can be a turning point. MAHA’s larger message has never been solely about preventing chronic disease; it is about protecting future generations. Healthier food, cleaner environments, and stronger metabolic health are not just wellness goals; they are fundamental fertility interventions.
If we are willing to address these upstream drivers with the urgency they deserve at the system and policy levels, we have an opportunity not only to avoid a national security threat posed by falling fertility rates but also to strengthen the health, resilience, and reproductive vitality of families for generations to come.
Related reading:







COVID SHOTS ARE THE REASON 4 POPULATION REDUCTION!!
One of the factors that wasn’t mentioned is the role that societal movements had. When JFK was President, the feminist movement was gaining ground.
Now, with technology and more influencers, there is more push to not have kids.
Here’s an article on the impact of feminism on society and ultimately fertility rates as well: https://unorthodoxy.substack.com/p/how-feminism-and-dei-destroy-humanitys