HHS Secretary Kennedy Promises Scientific Review of Peptides. Why That's a MAHA 'Win.'
Secretary Kennedy announced that the FDA will soon review a dozen peptides currently only available on the black market
On April 15, Health and Human Services Secretary Robert F. Kennedy Jr. took a giant step in the direction of legalizing peptides, promising on X to rigorously review a dozen peptides that former president Biden had classified as Category 2 – “Bulk Drug Substances that Raise Significant Health Risks - driving a dangerous black market.”
Kennedy wrote in his X post, “Today, we took long-overdue action to restore science, accountability, and the rule of law.”
Continued Kennedy, “Now, after nominators withdrew 12 peptides, the FDA will remove them from Category 2 and will bring them to PCAC [Pharmacy Compounding Advisory Committee] at its next two meetings, beginning in July—where independent experts will rigorously evaluate each substance on its scientific merits using full clinical, pharmacological, and safety evidence.”
After listing the 12 peptides the FDA will review during the PCAC’s summer meetings, Kennedy added, “This action begins to restore regulated access and will immediately begin shifting demand away from the black market. We will follow the science, enforce the law, and deliver the clarity patients, providers, and pharmacies deserve.”
Wednesday’s move tracks with Secretary Kennedy’s broader (and very MAHA) pledge to bring radical transparency to HHS and its sub-agencies. While some legacy media shot down Kennedy’s X post, castigating peptides as “unapproved therapies” used by influencers and celebrities, many MAHA supporters and proponents of medical freedom were ecstatic. “This is what we’ve been waiting for!!!” wrote Jesse Morse, M.D. on X.
For current users, legalization would be a blessing.
Every day, in bathrooms, bedrooms, and kitchens across America, thousands of people draw up compounds from tiny vials and inject themselves with substances with names like: BPC-157, TB-500, GHK-Cu, Ipamorelin, Semax, Selank, KPV, KLOW, Wolverine, and MOTS-c – many of which are on Kennedy’s ‘to review’ list.
Many users operate outside the medical system, finding suppliers online and ordering syringes and bacteriostatic water from Amazon while following dosing advice from influencers on Instagram or TikTok. What began in quiet corners of functional medicine has moved into homes across the country.
Why Did Peptide Demand Explode?
Peptides are short chains of amino acids that are signaling molecules in the body, regulating tissue repair, collagen production, appetite, and hormone activity. Synthetic versions of peptides are marketed for recovery, fat burning, performance, and healthy aging.
To many consumers, peptides are the fastest shortcut to becoming leaner, stronger, and more resilient — or at the very least, a higher-performing version of themselves. Users also believe peptides can help with hair growth, improve eyesight, increase cognition, accelerate fitness recovery . . . and the list goes on. Some people even believe peptides can make them superhuman.
While peptides have been around for decades, the market exploded with the arrival of GLP-1 weight-loss drugs. These drugs reshaped public awareness of peptide therapies almost overnight, while shortages and four-digit prices quickly followed.
How Peptides Went Underground
Those who wanted access to these ‘wonder drugs’ did not wait. Both compounding pharmacies and the so-called gray market moved quickly to meet the growing demand. That shift accelerated in 2023 when the FDA placed 19 peptides into Category 2, sharply limiting compounding while many of those products remained available online.
What followed was a peptide marketplace that now ranges from physician-guided compounding to direct-to-consumer gray-market sales and, at the far end, black-market products.
Compounding pharmacies can step in during FDA-recognized shortages and use formulation tweaks and other workarounds to keep meeting demand.
In a gray market, products are sold directly to consumers with companies using lab evaluations, certificates of analysis (COAs) and polished websites to seem more credible, despite operating outside normal medical channels. Black-market products are the cheapest, often sourced from China, Russia, or India, with uncertain purity, ingredients, and efficacy.
Cost drives much of this movement: Online gray market peptides can sell for $50 to $500 per vial, while physician-guided clinics can charge thousands per month for oversight and injections. Some peptides are FDA-approved prescription drugs, like GLP-1s, but many compounds promoted online are untested and sold as “research-only.” In practice, it means people have become their own guinea pigs.
The Fight Over Access
Prior to Wednesday’s announcement, Kennedy criticized a medical system that limits preventive choice, even as demand for these therapies, including peptides, is already here. When legitimate access narrows, people look elsewhere. Many go online, where access is easy, oversight is thin, and the risks are hard to judge.
A policy environment that restricts physician-guided access without addressing real demand does not solve the problem. It pushes more people into a gray market with fewer safeguards, less transparency, and more room for abuse.
Under Secretary Kennedy, that may soon change.
Social Media and the Peptide Rush
Search “peptides” on social media and there are hundreds of influencers telling viewers what to use, how to dose it, and where to buy it. Most of these figures do not have a medical background, and they do not steer people toward physician-guided care and compounding pharmacies. They send them to gray-market sellers, or outright black-market products. The market is shaped not by medicine and regulation, but by algorithms, affiliate links, and followers.
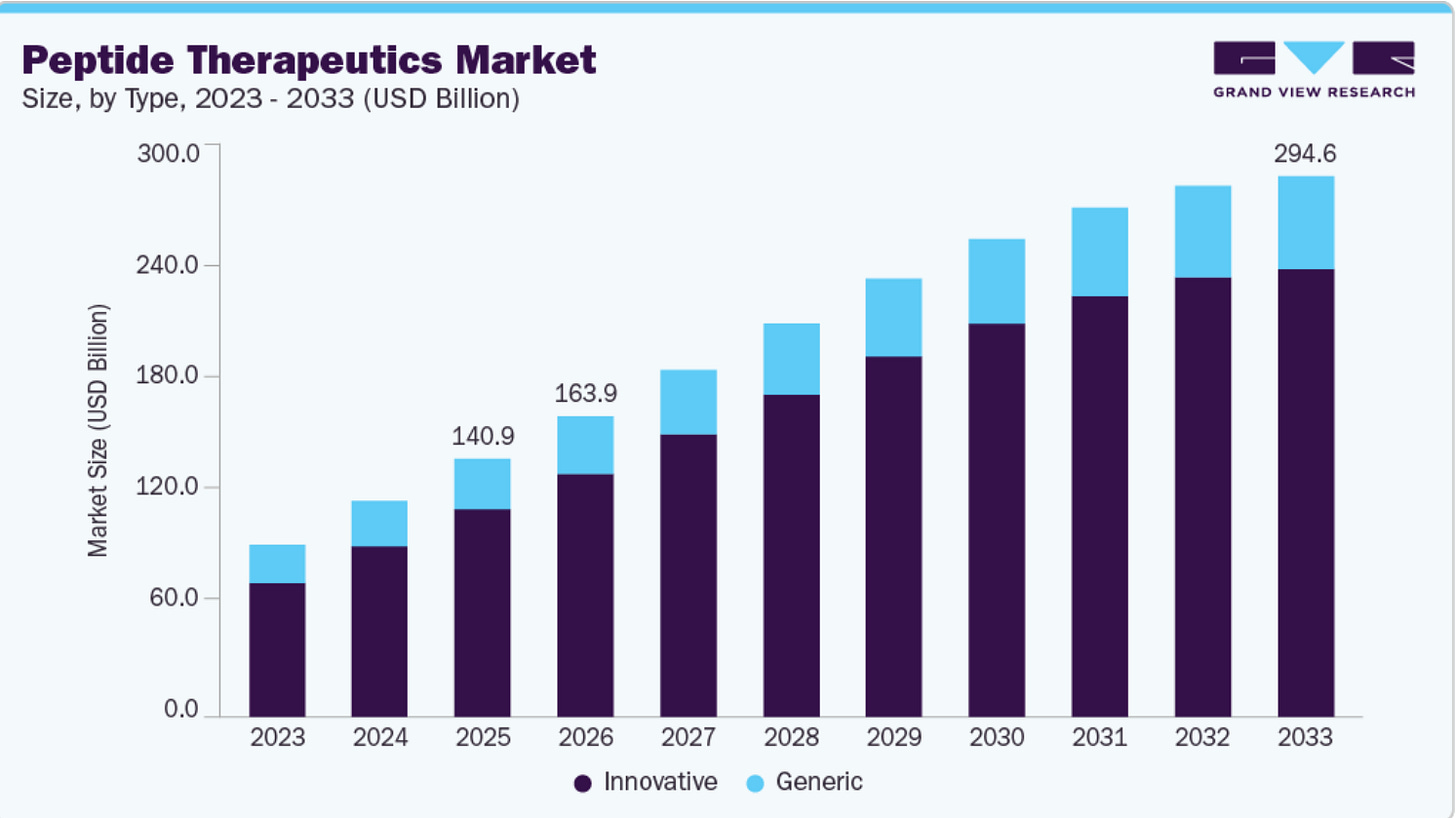
Source: Grandview Research https://www.grandviewresearch.com/industry-analysis/peptide-therapeutics-market
Peptides have become part of a much larger battle over access to preventive, out-of-pocket health care. This is no longer a niche market. By 2025, the global peptide therapeutics market was estimated at more than $140 billion, with projections reaching $294.58 billion by 2033.
That kind of growth tells its own story: demand for peptides and other wellness therapies is not going away. Americans want more options for healing, recovery, prevention, and longevity – and they deserve safe, transparent, medically supervised choices.
On The Joe Rogan Experience podcast in late February, Kennedy said he was a “big fan” of peptides and that the administration was moving to reverse part of the FDA’s 2023 peptide crackdown, which pushed 19 popular peptides into a category that blocked routine compounding by many pharmacies. Kennedy said roughly 14 of those peptides could soon be moved back into a category that would allow legal compounding under physician supervision.
Wednesday’s announcement makes it clear that the Secretary means business.
“My hope is that they’re going to get moved to a place where people have access from ethical suppliers,” Kennedy told Rogan, adding that HHS is “still looking at the science.” He framed the move as an effort to improve the quality of products people are already using.
The Right to Pay for Prevention
At its core, taking or not taking peptides is about health sovereignty and autonomy over one’s own health. Health-conscious Americans should not be forced into unsafe or unregulated channels simply because the medical system refuses to make room for legitimate preventive care.
As Secretary Kennedy recognizes, the answer is not to deny demand or drive it underground, but to make safer, transparent, medically supervised access easier than the gray market.
What happens after the PCAC’s summer meetings will determine whether peptides remain trapped between overregulation and the black market, or find their rightful place in a transparent, prevention-focused health system.






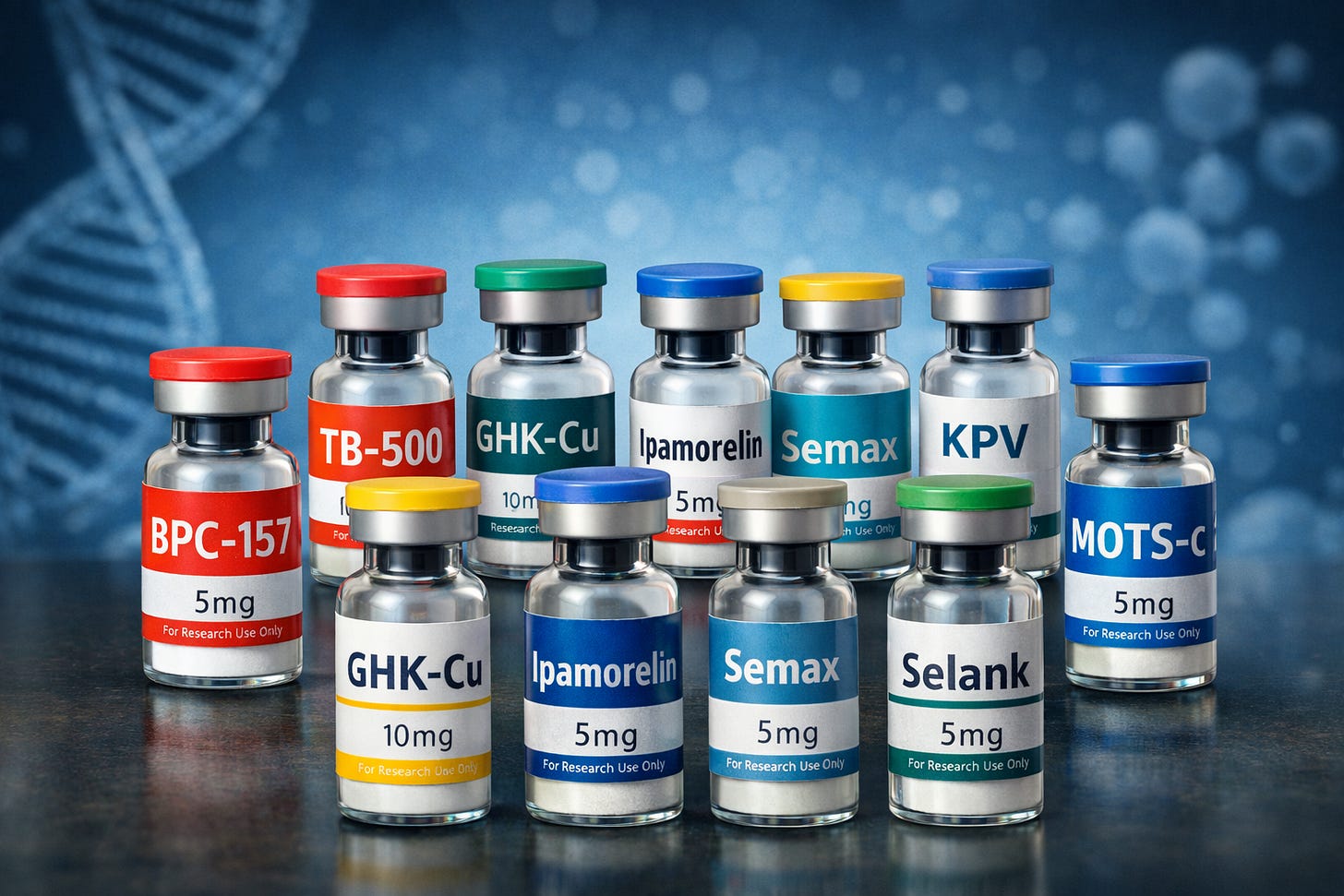


Peptides are a phenomenally useful technology that's made unnecessarily expensive and hazardous due to the FDA's forced grey market status. That's very healthy for pharma and very bad for the people. Hopefully RFK can get that turned around. Another problem is the FDA placing compounding pharmacies in a place where they have to worry about getting taken out of business for providing similarly safe health options. I recently wanted a compounded lotion formulation, and even though everything about it was simply over-the-counter nutritional ingredients, I had to have a doctor's prescription. So getting a $30 product would become a $300 product.
And as always: if RFK truly wants a healthy America, he HAS to address the poisons that we breath that are forced on us whether we want them or not, especially the fragrance industry. THIS is the shocking unregulated market that makes food additive downright wholesome. People who want to poison their clothes and themselves with fragranced dryer sheets should be forced by law to install catalytic converters on their dryers.
This is nothing but a sideshow distraction intended to divert public attention from the main issue, which is the COVID contagion and its deadly mRNA fake “immunizations” that were never intended to be anything but political mayhem, mass murder, and corrupt profits for “Big Pharma” that owns the congress and has corrupted and manipulated every aspect of medical practice and research, to the point of destroying medical science and productive research. The COVID contagion and its deadly fake immunizations were the worst abuse of medicine imaginable. They were a disguised form of mass murder and treason, for no country can defend itself with sick, crippled, dead and unborn soldiers and civilians. COVID and its fake immunizations were the product of military “germ warfare” research. I lost two cousins to this atrocity. One was a board certified anesthesiologist who was murdered by injection in the guise of a requirement for working in her hospital. The other was a NASA policeman who caught COVID along with his wife when they flew from Florida to Mississippi to visit relatives during the contagion. The wife survived, but he made the mistake of going to a hospital where he was murdered by mechanical hyperventilation, which offers no benefits and is inherently harmful because it depletes body reserves of carbon dioxide and undermines lung function and tissue oxygenation. Big Pharma has grown rich and powerful courtesy of lavish government subsidies that were never justified. It uses mass media saturation advertising to brainwash citizens as the backbone of its marketing strategy. The government courts have sanctified this criminal activity on the grounds of “free speech.” Since when is “free speech” a legalized means of promoting this criminal activity? Why is this prohibited in every country EXCEPT the United States???? Big Pharma is viciously manipulating the courts and state legislatures and medical boards and journals purely for the sake of profit at the price of public health. Government in the United Socialist States of what used to be America has become the TYRANNY that it was intended to prevent. It is an abject failure. It serves only to protect and promote corrupt corporate profits.